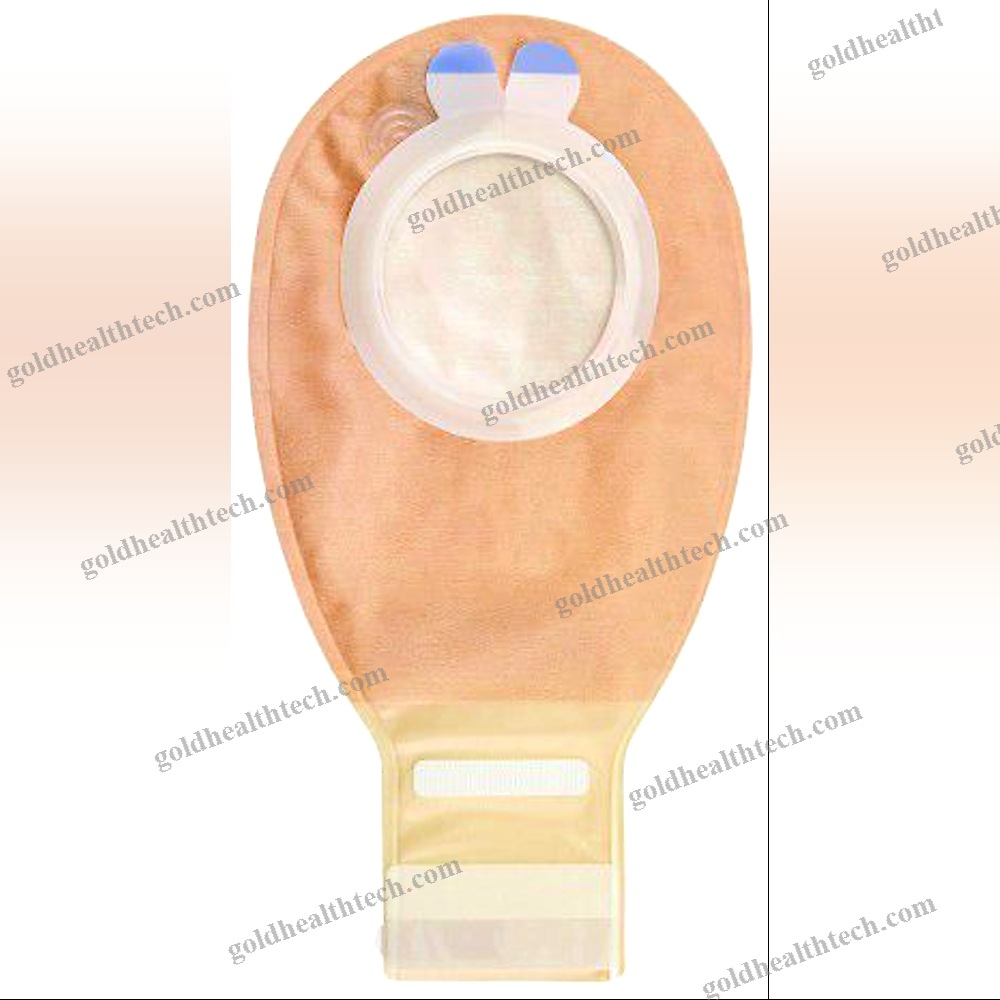
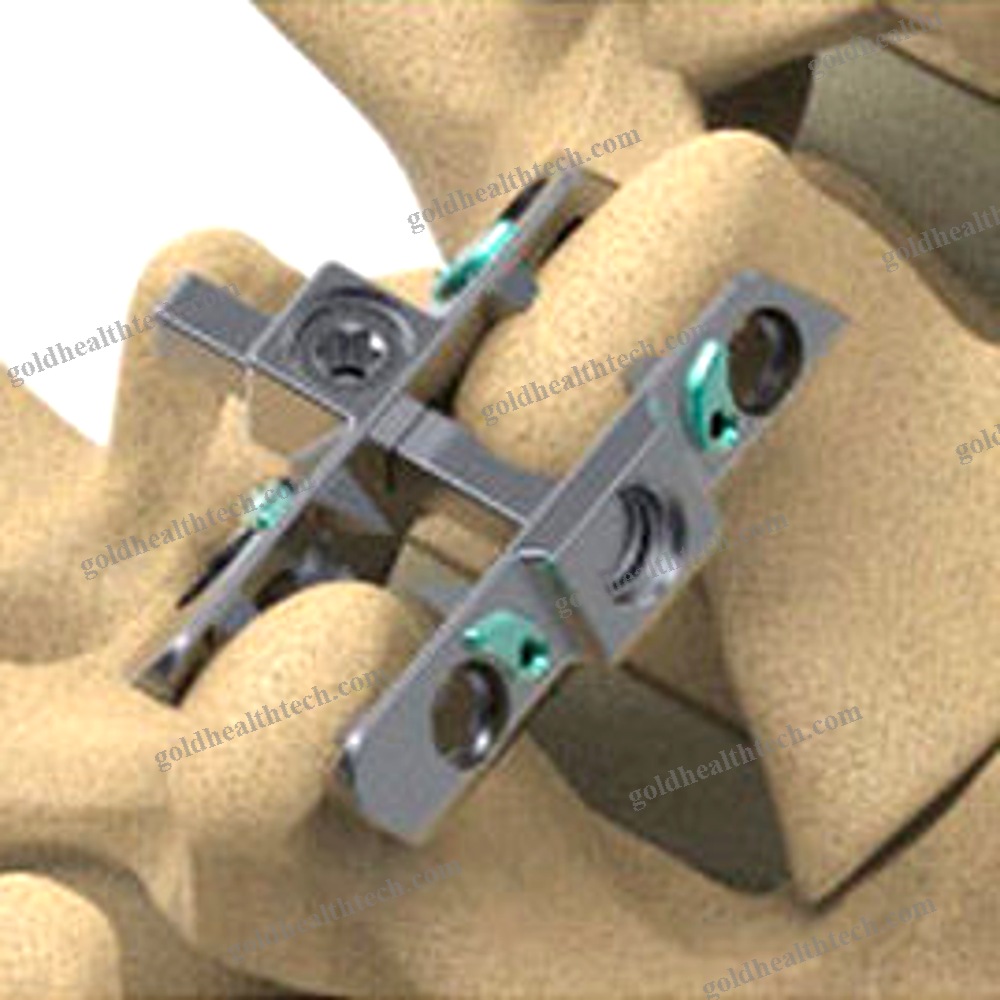
Lumbar Interspinous Vertebral Implant
| Spinal Section | Lumbar, Sacral, Thoracic |
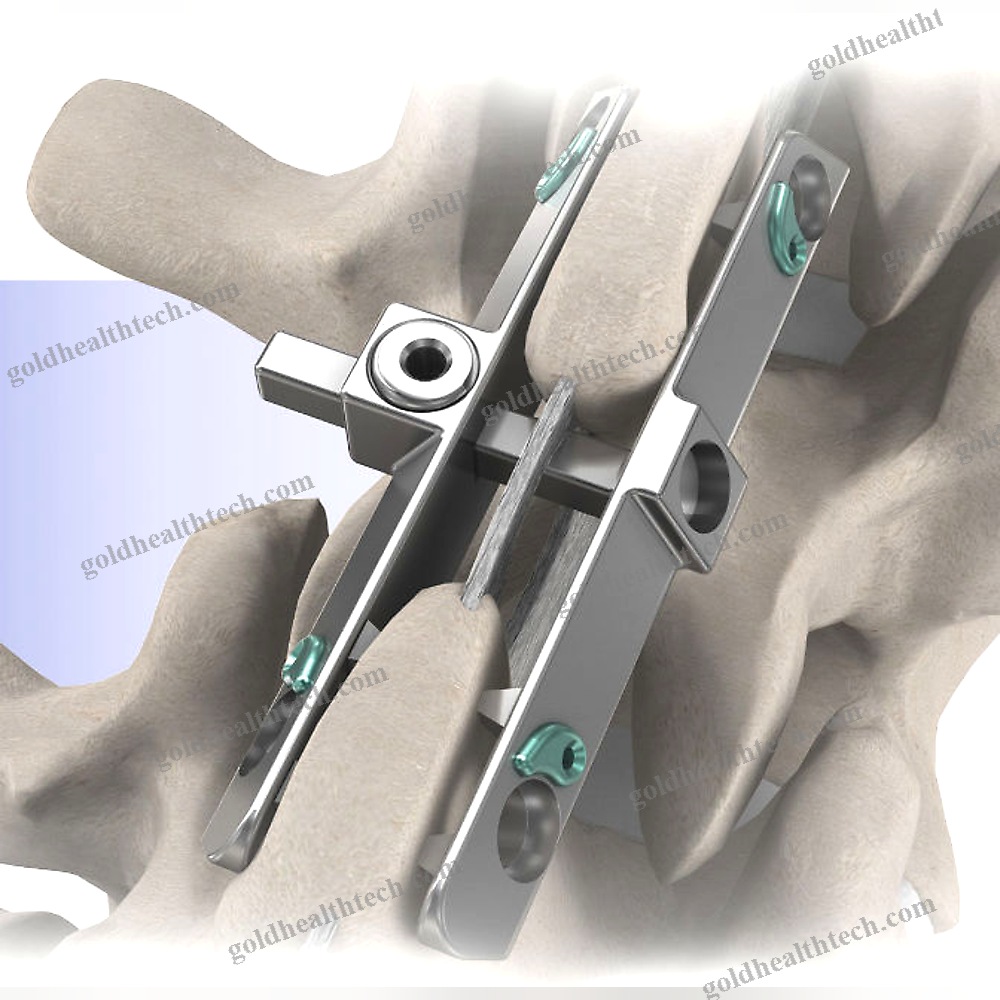
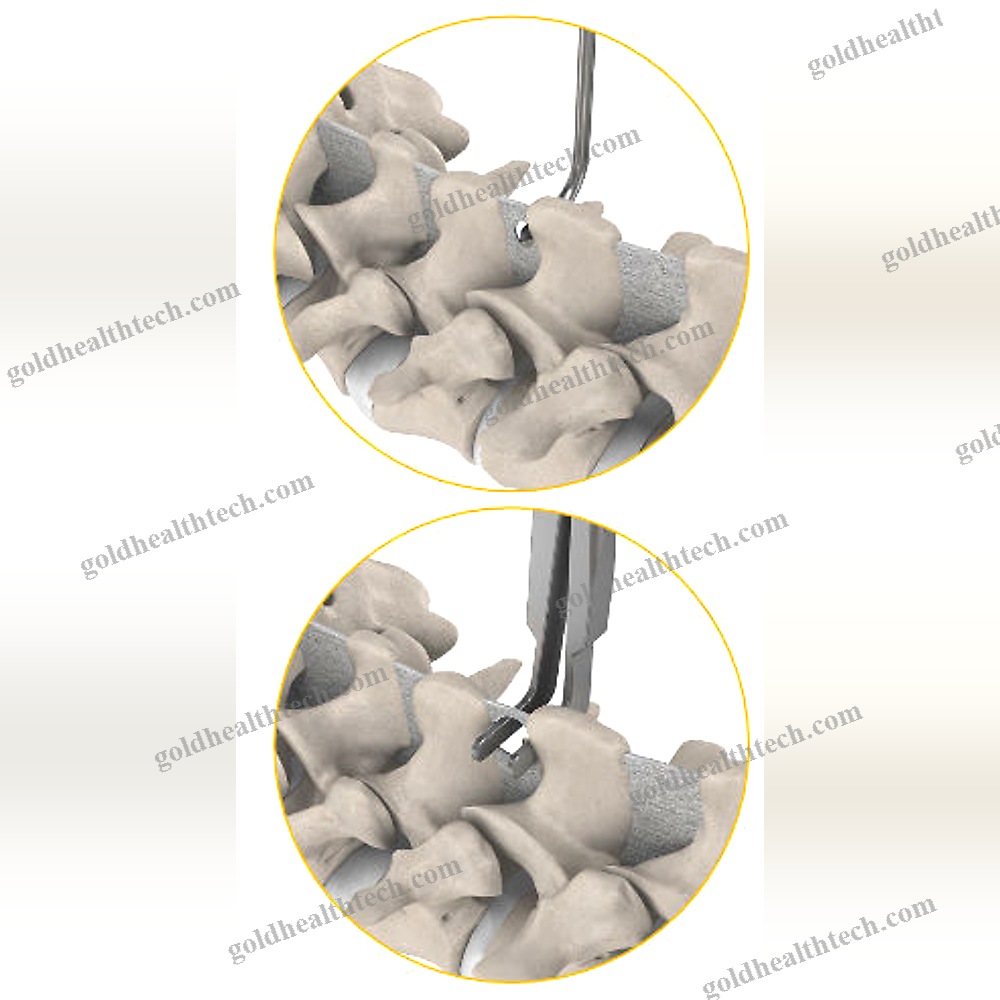
The Reli™ SP PLUS Spinous Plating System is the first spinous plating system to receive 510(k) marketing clearance from the U.S. Food and Drug Administration (FDA) for use with supplemental screw fixation. The two-plate design includes an unparalleled fixation option that enables surgeons to attach the plates to the spinous process using screws in addition to cleats. This construction is more rigid, which reduces the likelihood of occult spinous process fracture and mitigates against dislocation migration.input:
output:System capabilities:The initial product to obtain FDA clearance for use with supplemental screw fixationPyramidal Cleats and Supplemental Screw Fixation establish a more rigid framework.
Reduces the likelihood of occult spinous process fracture.Reduces the risk of dislocation migration
Four plate sizes are available to ensure an optimal anatomical fit.


MAECENAS IACULIS
Vestibulum curae torquent diam diam commodo parturient penatibus nunc dui adipiscing convallis bulum parturient suspendisse parturient a.Parturient in parturient scelerisque nibh lectus quam a natoque adipiscing a vestibulum hendrerit et pharetra fames nunc natoque dui.
ADIPISCING CONVALLIS BULUM
- Vestibulum penatibus nunc dui adipiscing convallis bulum parturient suspendisse.
- Abitur parturient praesent lectus quam a natoque adipiscing a vestibulum hendre.
- Diam parturient dictumst parturient scelerisque nibh lectus.
Scelerisque adipiscing bibendum sem vestibulum et in a a a purus lectus faucibus lobortis tincidunt purus lectus nisl class eros.Condimentum a et ullamcorper dictumst mus et tristique elementum nam inceptos hac parturient scelerisque vestibulum amet elit ut volutpat.